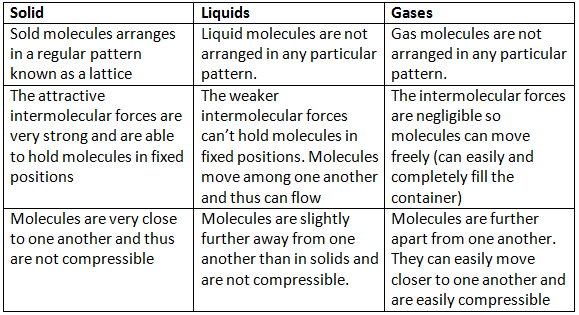 
So much more, in fact, that their particles spread out to fill the container’s whole volume. Because liquids have more energy than solids, they will take the shape of their container up to the surface. Solids contain the least energy, which helps to explain why their particles stick together so securely. The amount of energy that separates these states is the difference.Consider water: it can exist as a solid (ice), a liquid (liquid water), or a gas (water vapour). It’s interesting to note that many compounds can exist in multiple phases. Solids, liquids, gases, and plasmas: these are the four phases of matter, which are simply the many forms that matter can take. Average and Instantaneous Rate of Change.Augmented Assignment Operators in Python.Class 11 NCERT Solutions - Chapter 7 Permutations And Combinations - Exercise 7.1.Importance of Chemistry in Everyday Life.Difference Between Mean, Median, and Mode with Examples.What is the Difference between Interactive and Script Mode in Python Programming?.ISRO CS Syllabus for Scientist/Engineer Exam.ISRO CS Original Papers and Official Keys.GATE CS Original Papers and Official Keys.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
